|
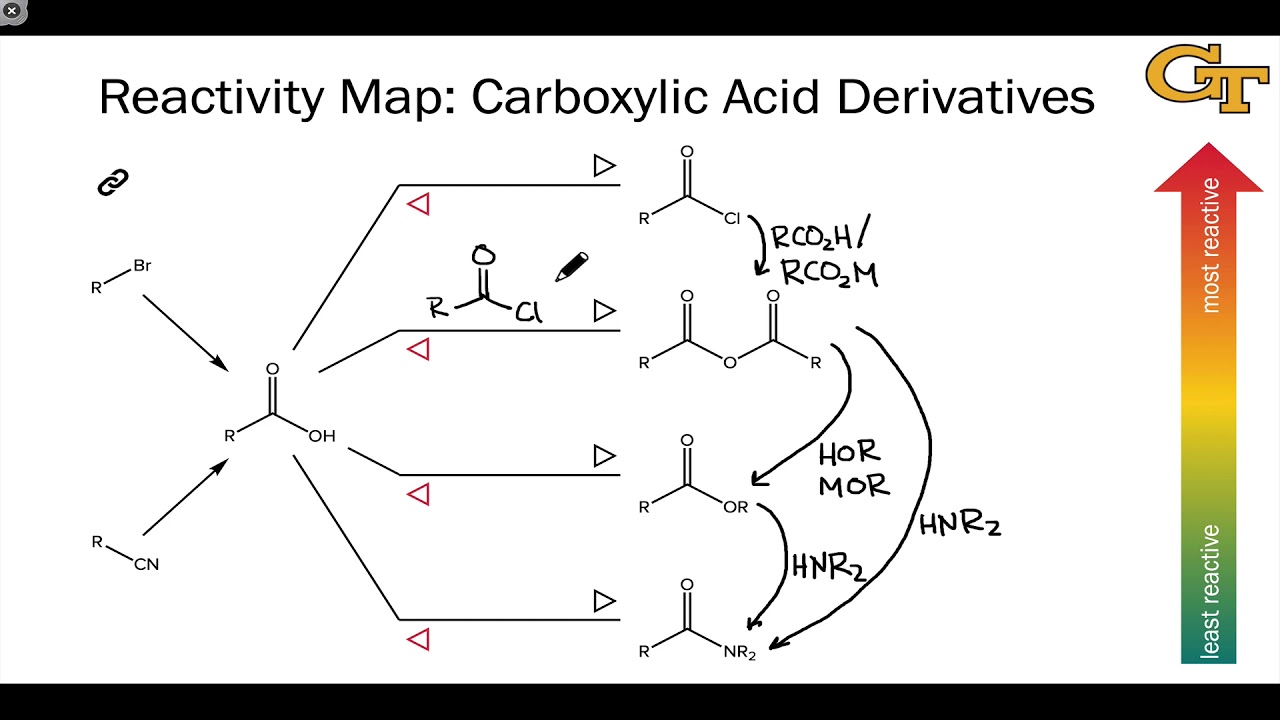
Strain: An increase in molecular potential energy due to electron repulsion or a deviation from ideal geometry. The following sections detail how the various carboxylic acid derivatives can be converted one into another. 
S teric hindrance: The prevention or retardation of inter- or intramolecular interactions as a result of the spatial structure of a molecule. Carboxylic acid derivatives are very reactive. Steric effects complement electronic effects, which usually dictate shape and reactivity. Steric effects result from repulsive forces between overlapping electron clouds. Steric effects: Nonbonding interactions that influence the shape (conformation) and reactivity of ions and molecules. The term stereoelectronic effect is also used to emphasize the relation between the electronic structure and the geometry (stereochemistry) of a molecule. This gives an overall effect of substitution, and is the mechanism of the common nucleophilic acyl substitution often seen with esters, amides, and related structures.Įlectronic effect: Influences the structure, reactivity, or properties of molecule but is neither a traditional bond nor a steric effect. Nucleophilic addition-elimination: A two-stage reaction process of an addition reaction followed by an elimination reaction. S teric hindrance: the size of the substituent affecting a reaction’s ability to proceed
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
